Update 3 December: The study is ongoing and actively recruiting participants. More information is available on the study’s own website.
On September 5, the Swedish Medical Products Agency approved our new clinical migraine study. Recruitment of participants has now started via inmestmottagningen.se, and we expect the study to begin in October.
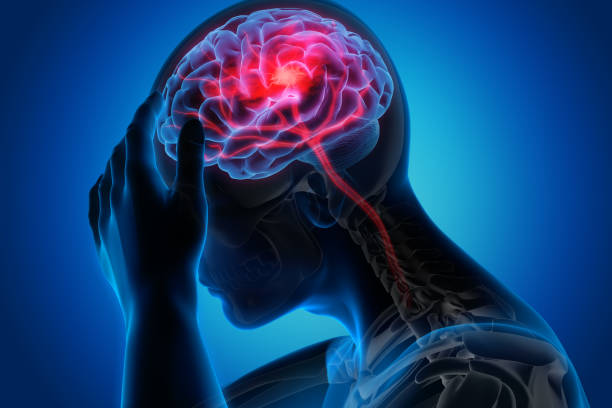
The study targets adults with migraine and aims to investigate whether INMEST, previously tested for other indications, can reduce migraine frequency and improve quality of life. Participants will use the treatment at home for six weeks, where mechanical stimulation inside the nose activates both the trigeminal and vagus nerves — two key components involved in migraine mechanisms.
A total of 110 participants are planned for this open clinical study. The study lasts up to 20 weeks per participant, including observation periods and six weeks of active treatment for all. Effectiveness will be evaluated using diary data, migraine-specific questionnaires, and patient-reported outcomes. Participants will need 4–5 clinic visits; the rest of the study is conducted at home.
The study is conducted by Abilion Medical Systems in collaboration with the INMEST Clinic and is funded by the INMEST Foundation. We look forward to contributing new knowledge on how nerve stimulation can be used in migraine — an area with a strong need for new treatment options.
If you are interested in participating, please read more on the study’s information page (in swedish). You can register your interest via email at [email protected] or by calling the clinic at +46 (0)8 414 005 90.
For any questions about the study, please contact the INMEST clinic directly